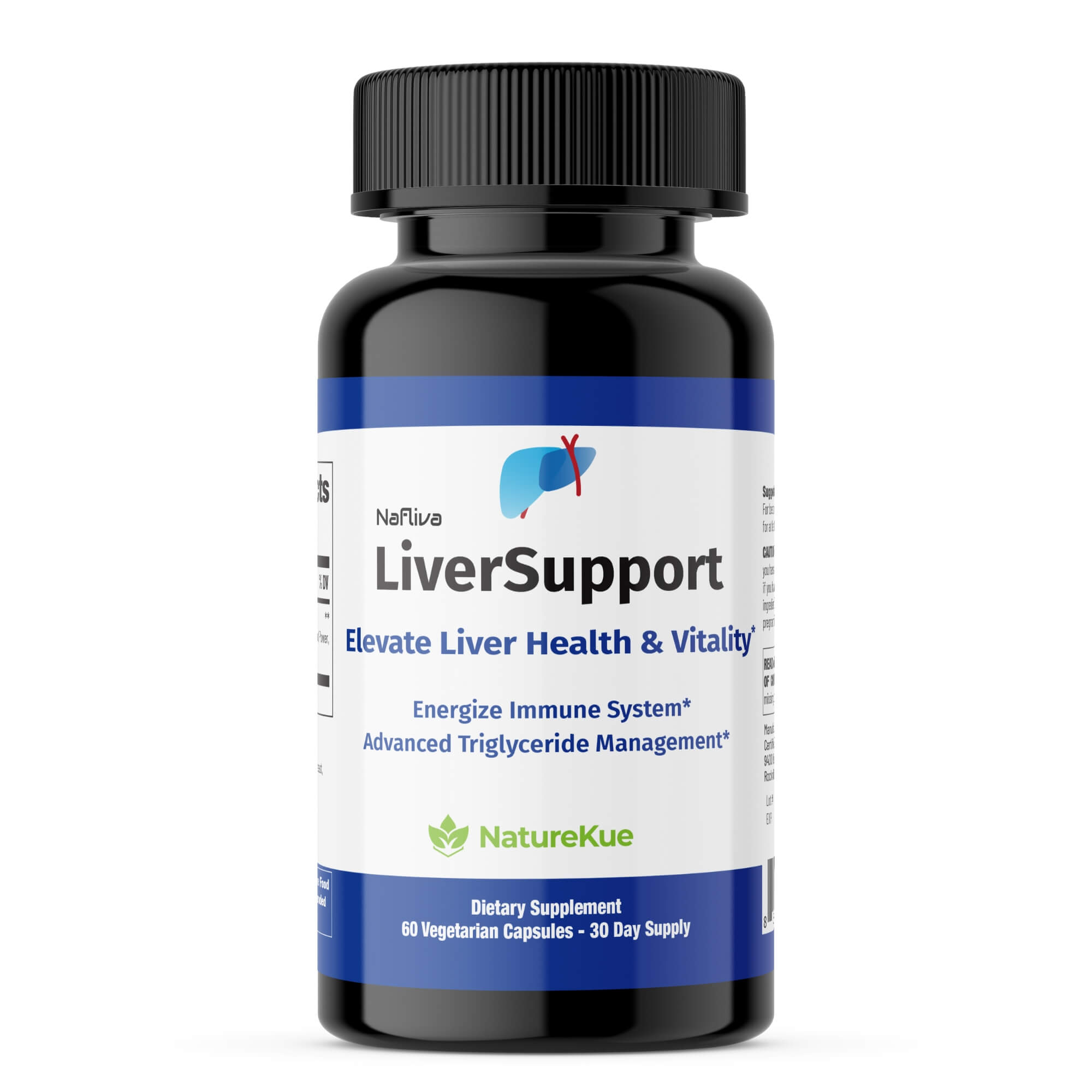
Boost Health & Vitality with NatureKue
NSF cGMP certification and GMP for Sport accreditation.
Dietary Supplements
View allAll-In-One Fats Campaign
NatureKue Metabolism
Elevate your daily health regime with this powerful supplement, essential for safely weight management and healthy metabolism of fats and proteins*
A Natural Solution for Long-Term Well-Being